November 04, 2017
Since 1961, meat consumption has grown almost twice as fast as the human population (FAOSTAT 2013), and as a result, the total biomass of animals raised for food now outweighs that of humans and wildlife combined. This simple observation illustrates the profound transformations that have occurred in the food production sector to support our growing appetite for meat. Amongst the many technological innovations that have enabled the intensification of animal farming is the routine use of antimicrobials. In many countries, veterinary antimicrobials have become imperative to maintain health and productivity in densely populated farms. In 2013, 73% of all antimicrobials sold on earth were used in animals, mostly as growth promoters, or as surrogates for good hygiene measures that could otherwise prevent infections. This expanding use of antimicrobials in animals has been a subject of considerable concern in light of the threat of antimicrobial resistance (AMR).

In a recent peer-reviewed policy forum published in Science, researchers from CDDEP led the first comparative assessment of global measures that could help reduce antimicrobial use in animals. The objective of this work was twofold: first, comparing the order of magnitude of different options to cut antimicrobial use to distinguish realistic policies from anecdotic measures; second, to start a discussion about the range of options available to cut antimicrobial use, and identify the areas of research where further work is needed to translate those options into applicable policies.
The first approach, which proposed cutting antimicrobial use (-66%) by limiting meat consumption to 40 grams per day is admittedly ambitious. However, it would carry health benefits other than solely reducing AMR, and more importantly, it provides us with an estimate of the effort to accomplish this through other measures if we wish to keep consuming meat at current levels. Those efforts could be proportionally reduced if a reduction in meat consumption was included as part of a combination of measures to cut antimicrobial use in animals.
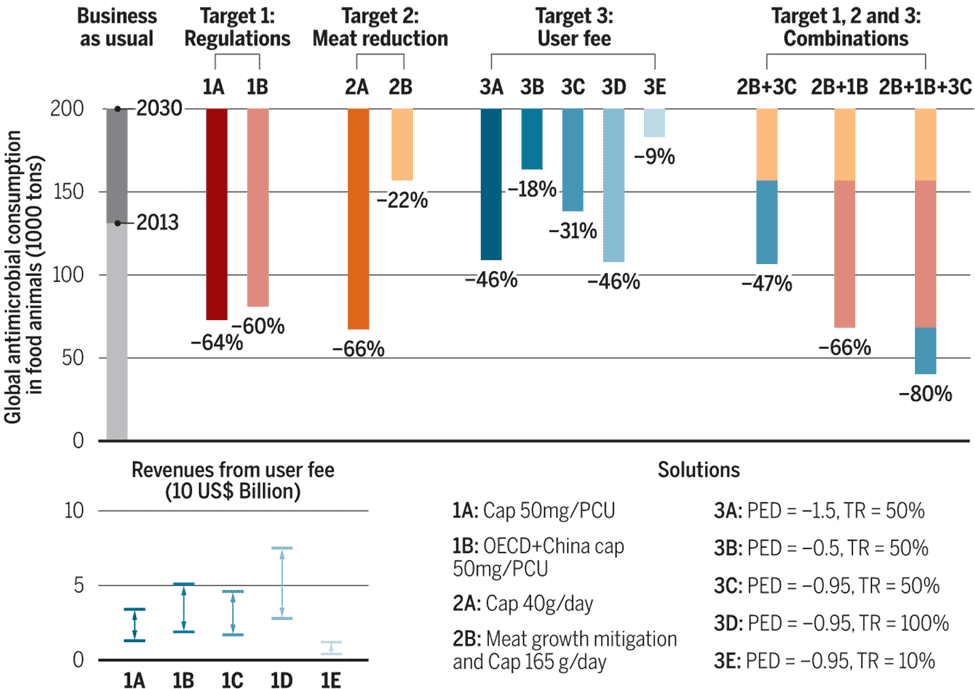
The second approach, which proposed to introduce a regulation to cap antimicrobial consumption at 50 milligrams per kilogram of meat produced, seems more feasible (-64%). First, because a handful of countries have demonstrated that it is credible to operate a profitable livestock sector under this consumption target. Second, because by its simplicity this measure could, in theory, reduce antimicrobial use to a predictable amount in each country. However, a major limitation of such a policy is that it relies on enforcement and surveillance capacities. In low- and middle-income countries (LMICs) where veterinary services are limited, inspecting millions of farms each year to control antimicrobial use would be an incredible challenge. But it is precisely in those countries that antimicrobial consumption is growing the fastest to support the growing demand for meat consumption. In some high-income countries, enforcement could also be problematic given that veterinarians are both prescribers and providers of antimicrobials.
A third approach, introducing a user fee on veterinary antimicrobials, may help circumvent those limitations. Concretely, compared with the first two options, it appears more feasible to impose a 50% fee at the point of manufacture or import because of the limited number of antimicrobial manufacturers. However, this would only lead to a 31% reduction in antimicrobial consumption. In addition, a user fee policy would need to be adapted to ensure that LMICs are not disproportionally affected by the fee. In particular, treatment needs to remain affordable for farmers who rely on the ability to treat their animals for subsistence.
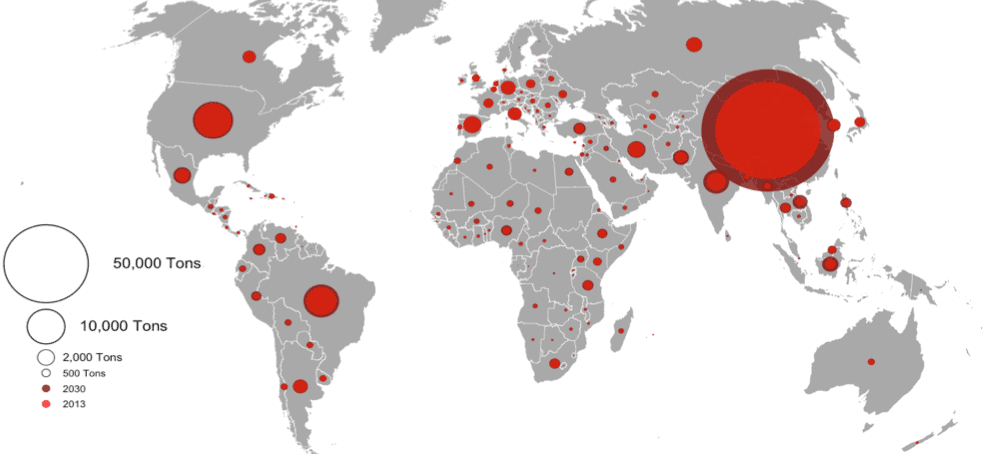
For this assessment, we also projected the global consumption of antimicrobials in food animals for 2013 (131,109 tons) and 2030 (200,235 tons). Our projections are an alarming revision of a study published two years before in PNAS that initially reported the global consumption at 66,000 tons in 2010. So, what happened? This large increase mostly reflects better reporting: new reports have shown that China is the largest consumer of veterinary antimicrobials in both relative and absolute terms, and this has inflated the global consumption figures between 2010 and 2013.
In 2013, in relative terms, the average global amount of antimicrobials used per animal was estimated at 118 milligrams per kilogram. However, considerable variations exist between countries; the global average consumption by country was 50 mg/Kg, but in China this could go as high as 318mg/Kg while in the more advanced nations in the fight against AMR, such as Norway, the national average was only 8 mg/Kg. Unfortunately, if efforts to curb AMR remain uneven, its consequences will be shared by all, and the progress achieved amongst others in Europe will stand in jeopardy.
This work was also the opportunity to compare antimicrobial consumption in animals and humans. Numerous metrics have been proposed to measure antimicrobial consumption in animals and each has its practicalities (laboratory setting, clinical setting, etc.). However, as long as a consensus is not adopted across all countries, simple metrics such as the weight of active ingredients, will rely on less error-prone metrics to compare antimicrobial consumption. On this basis, we identified that humans and animals use roughly the same amount of antimicrobial per unit of biomass, 133 mg/kg vs 118 mg/kg (Klein et al., submitted).
This has two important implications. First, is it morally justifiable to allocate our pool of antimicrobial effectiveness evenly between humans and animals raised for food? Shouldn’t antimicrobials be allocated in priority to humans, where their use could avert hundreds of thousands of deaths due to the lack of access to treatment? Second, new drug-resistant mutations are more likely to occur in the animal reservoir because while humans and animals use approximately the same amount of antimicrobials, the biomass of animals by far exceeds that of humans. Thus, even if–as acknowledged in our study–the evidence for the transfer of resistance pathogens is not uniform for all pathogens, the animal reservoir has greater potential for the emergence of new resistance genes. These genes can subsequently be transferred to human-adapted pathogens. In a recent study, it was suggested that this may indeed be the dominant mechanism for the transfer of resistance, but also the hardest to quantify.
In conclusion, the intensification of animal farming has enabled the production of affordable sources of protein through substantial economies of scale. But, raising billions of animals for food comes at a cost for our health and the environment. At present, the cost associated with this loss of antimicrobial efficacy is not reflected in the price that consumers pay for meat produced with antimicrobials. Farmers and customers should bear the full costs for their choice of means of production; introducing a user fee on veterinary antimicrobials enables those costs to be internalized in the decision process.
*Thomas Van Boeckel is a post-doctoral visiting scholar at CDDEP and fellow at the institute of integrative Biology at ETH Zurich, Switzerland. ([email protected])