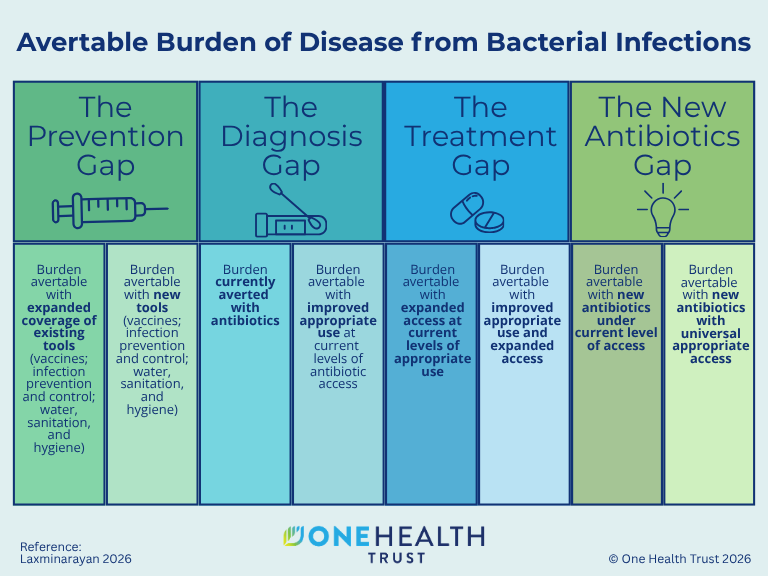
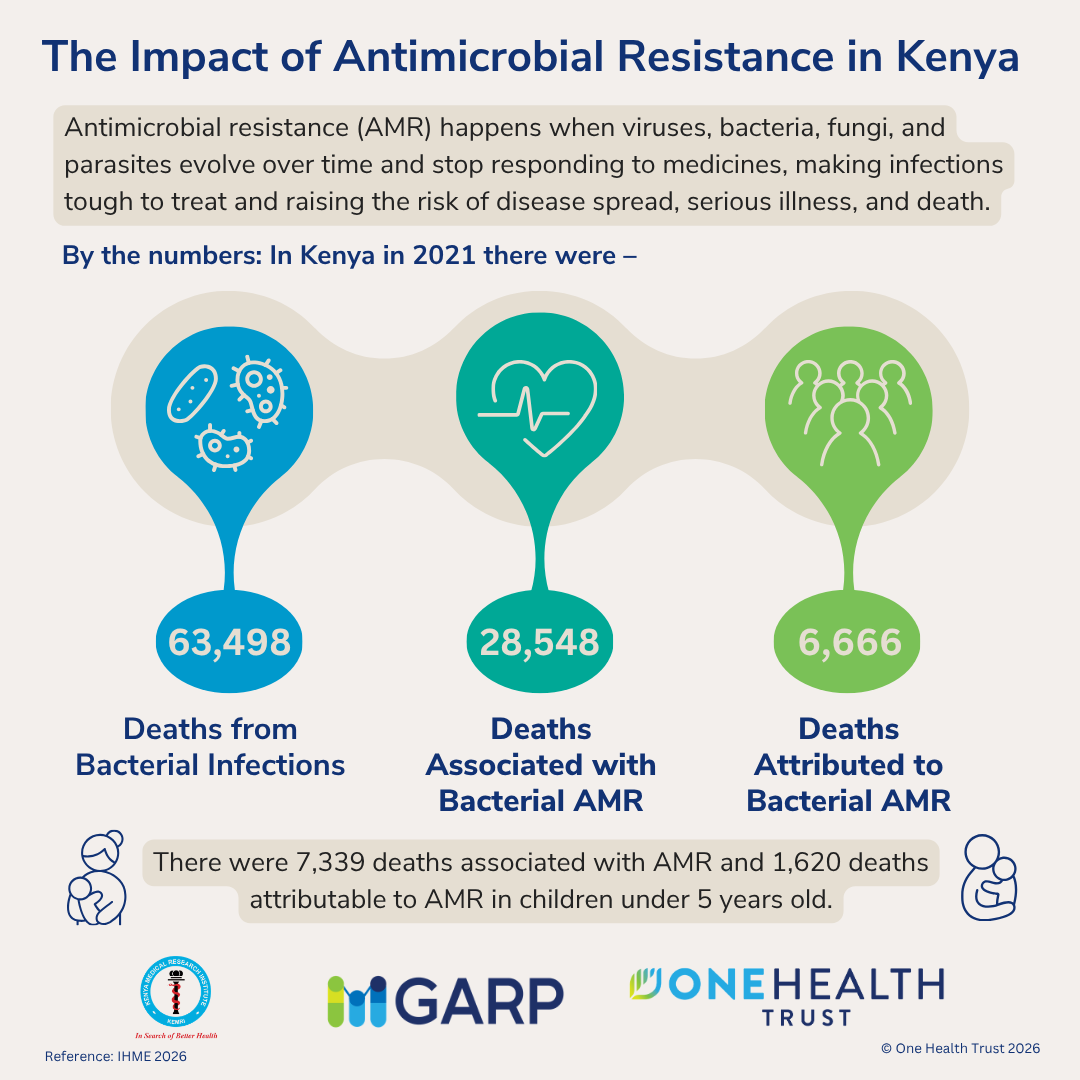
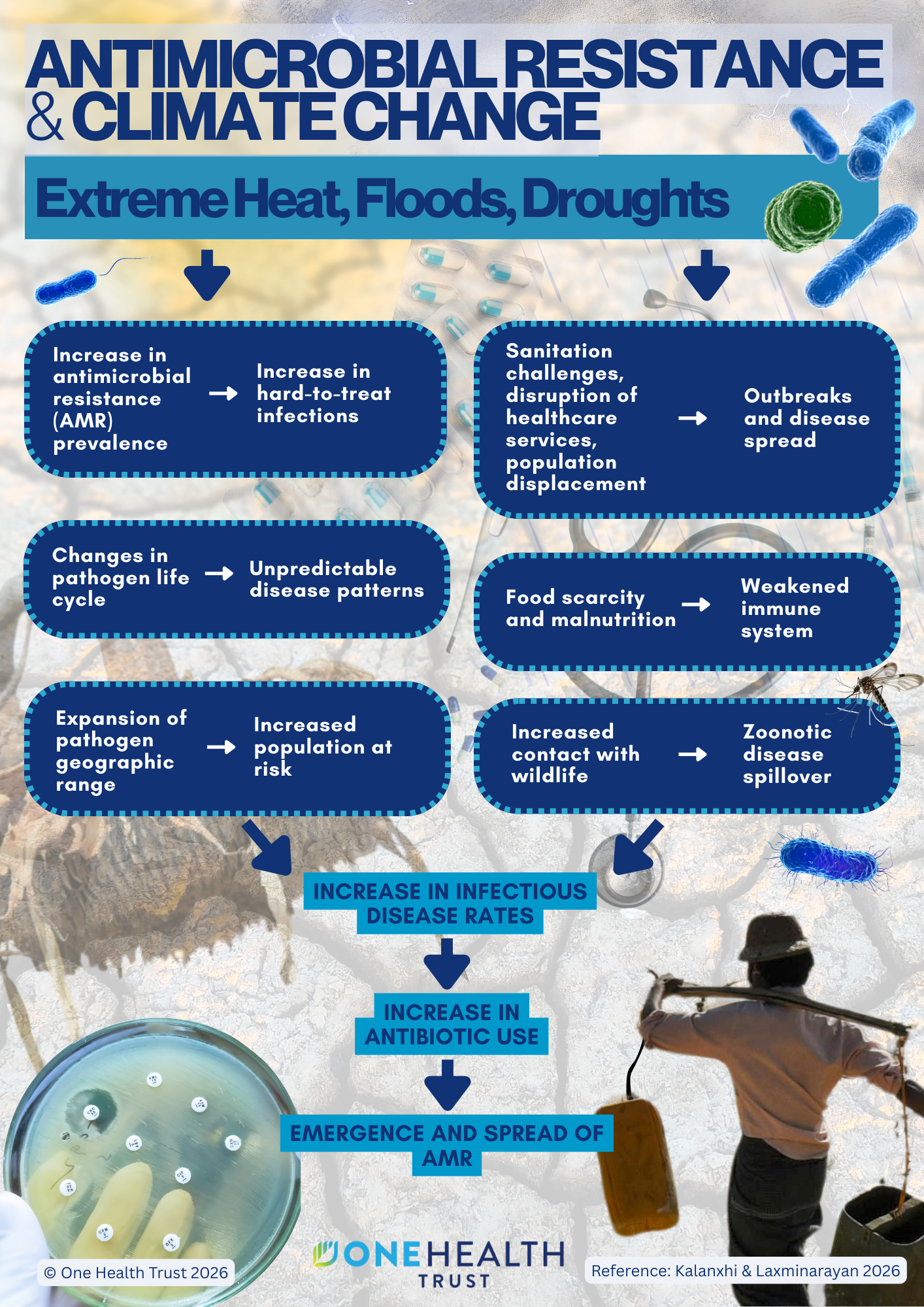
October 16, 2014
In the United States, creating laws and policy through Congressional legislation is the key method for regulating issues that are affecting the American population — including critical issues like antibiotic use and resistance. As antibiotic resistance has risen over the past decade, Congress has sometimes recognized the need for new laws and regulations to guide human and agricultural antibiotic use. Proposed regulations pertaining to antibiotic resistance vary from research and education grants on resistance to market incentives for new antibiotics to comprehensive plans for limiting antibiotic use in animals.
Using data from CDDEP and Extending the Cure legislative roundups and proposed and passed legislation from the Senate and House of Representatives from the 108th to 113th Congresses, this timeline shows bills that have been introduced in the last decade, for which use they are targeted, and their furthest progression in Congress. Though 80% of antibiotic use in the US by weight is related to agriculture, only 5 out of 14 bills introduced in this time period relate to farm and agricultural use. And despite a wealth of evidence that throughout this time period antibiotic resistance is rising and FDA antibiotic approvals are declining, only 2 of the 14 bills were eventually signed into law. One of these bills — the Antibiotic Development to Advance Patient Treatment (ADAPT) Act — is currently being reviewed by the House Committee on Energy and Commerce health subcommittee, which held a hearing in September to dicuss the legislation.